
Theme 4: Raising the standards
The Northern Health and Social Care Trust is committed to raising the standards by putting in place robust and meaningful standards against which performance can be assessed, involving service users, carers and families in the development, monitoring and reviewing of standards.
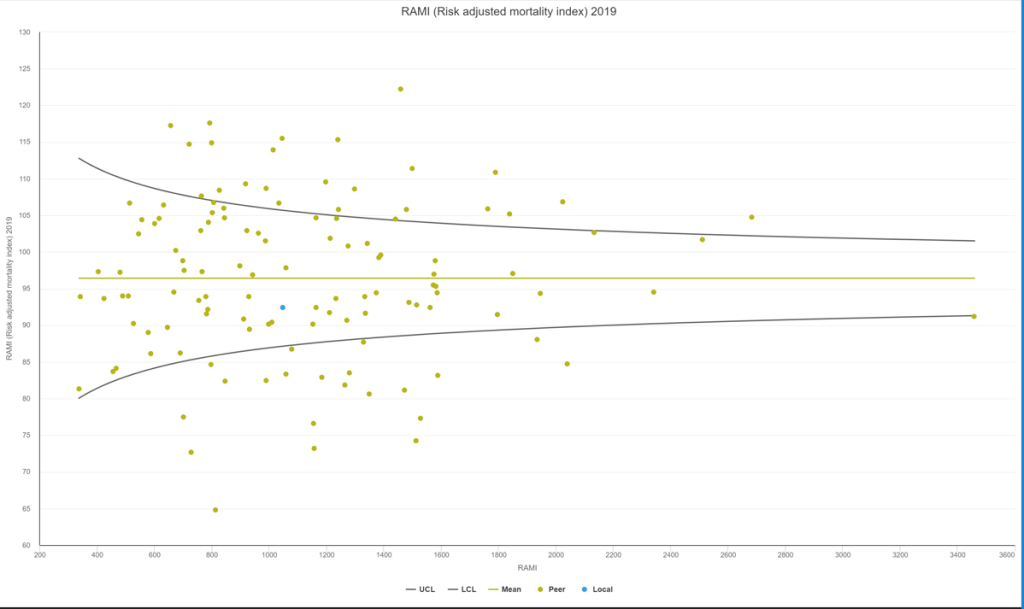
Risk Adjusted Mortality Index
The Risk-Adjusted Mortality Index (RAMI) is calculated by an independent healthcare benchmarking company called Caspe Healthcare Knowledge Systems, Ltd. Based on the age and condition of patients in the Trust’s hospitals, it calculates how many patients died compared to how many would be expected to die. The expected number of deaths is calculated using National Health Service (NHS) digital data which is based on the 2019 RAMI Model. A RAMI of 100 means that mortality was exactly in line with expectations; over 100 means more deaths occurred than would be expected, and below 100 means fewer than expected deaths. The Trust’s RAMI for 2023/24 (excluding palliative care patients) was 91.27. Note – Trust COVID-19 related deaths are excluded as no COVID-19 related deaths were included in the baseline used to create the index.
The chart shows the Trust’s RAMI (blue dot) compared to a cohort of English acute Trusts (one green dot per Trust). The further a dot is to the right, the more patients the Trust treated during the year, and the higher up the chart, the higher the RAMI. Dots within the funnel are inside the normal limits of variation. The Trust’s RAMI is below 100 and within the normal limits of variation, providing assurance the Trust is providing safe care to its patients.
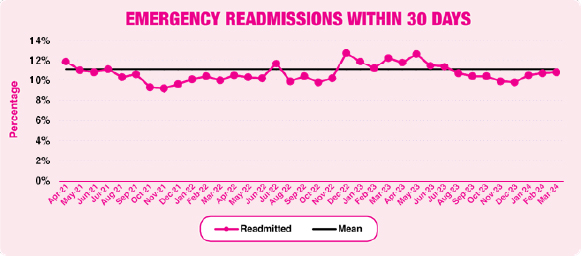
Emergency readmission rate
The 30 day readmission rate is a healthcare quality metric that measures the percentage of patients who are readmitted to a hospital within 30 days of being discharged from an initial hospital stay. The average readmission rate in 2023/24 remained the same as the previous year.
Emergency Department (ED)
The following table shows Antrim and Causeway performance for the past three years for:
- Total number of attendances
- Percentage of patients seen and admitted or discharged within 4 hours of arrival at ED
- Number of patients spending more than 12 hours in ED
- Percentage of patients seen by a clinician within 1 hour of arrival
- Percentage of patients who did not wait to be seen
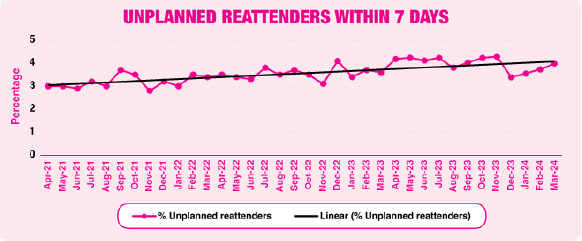
- Percentage of patients who re-attended within 7 days with the same complaint
| Site | Year | Attendances | 4 hrs | 12 hrs | Seen <1 hour | Did not wait | Reattenders |
| Antrim | 2021/22 | 91,041 | 54.30% | 10,157 | 30.24% | 3.77% | 3.25% |
| 2022/23 | 94,900 | 45.69% | 14,568 | 20.44% | 5.78% | 3.14% | |
| 2023/24 | 100,163 | 40.02% | 17,078 | 16.70% | 6.92% | 3.63% | |
| Causeway | 2021/22 | 45,434 | 60.31% | 3,764 | 22.19% | 5.16% | 3.05% |
| 2022/23 | 46,997 | 54.13% | 5,911 | 21.96% | 6.16% | 4.39% | |
| 2023/24 | 49,694 | 51.72% | 6,164 | 21.88% | 6.11% | 4.70% |
The following chart shows the percentage of people who re-attended within 7 days:
Actions taken to improve standards
Both Emergency Departments have remained challenges over 2023/24 with high decisions to admit remaining in both EDs for a prolonged period. An increased footprint has been provided on Antrim site to mitigate and maintain the 4-hour target but despite this increased space, the targets remained challenged due to demand. Work is ongoing with NIAS colleagues to maximise the ambulance turnaround while new pathways to avoid ED attendance are being launched across a number of specialties.
No More Silos
An expansion of the urgent care pathways on both sites has been undertaken with a new Acute Ambulatory Hub opening on Causeway Site which has provided the acute medical team the space to ambulate suitable patients, reducing the need for inpatient stay.
Reducing the risk of hyponatraemia
The Trust continues to participate in the regional implementation programme in response to the 2018 Inquiry into Hyponatraemia-related Deaths (IHRD). Following the closure of phase 1 of the programme in December 2022, the Trust IHRD task and finish group has worked towards implementation of phase 2a and 2b of the programme, with 27 of the 37 actions relating to the Trust complete, and the majority of the remainder paused whilst awaiting further regional guidance on Duty of Candour/Being Open.
Single registrant pre-administration checks of a blood component
Research carried out in the field of Transfusion has shown that a single person check before administration of a blood component is as safe as two people carrying out the check. It is considered that when a single person carries out the pre-administration check they are more focused, less likey to be interrupted and distracted and more likely to contact blood bank if there are any discrepancies during the checking procedure. In addition, single checking could be viewed as a time-saving measure and be more of a help to staff than having to locate someone else to check patient details with. Furthermore, the second staff member is not interrupted from their care of other patients to carry out the check.
Evidence from the Regional Haemovigilance Team has revealed that Western Health and Social Care Trust and Belfast Health and Social Care Trust are both using this in a number of areas and feedback has been very positive. Community Transfusion teams within Northern Health and Social Care Trust are also well used to this process and have been successfully carrying out single pre-administration checks for several years.
As per Trust policy, pre-administration checks of a blood component can now be carried out by one registrant. This practice continues to be rolled out across Antrim and Causeway sites by Haemovigilance, although further progress has been paused until Encompass has been rolled out. All areas within the medical division (with the exception of ED) are carrying out single registrant pre-administration checks of a blood component. Work has commenced with the surgical division with successful pilots underway in Wards C3 and C4, Wards Surgical One and Two in Causeway and the Surgical Elective Unit in Antrim. If all continues to go well, the plan is to move to Theatre, Day Procedure Units and Day Surgery Units.
Haemovigilance have been providing ward educational support sessions to staff who are feeling anxious about the changes with continued success and offer weekly check-in sessions with ward managers and deputy ward managers. To date no issues have been identified and progress continues with reported benefits for patients and staff. Monitoring of single pre-administration checks of blood components by Haemovigilance, has not shown any increase in incidents reported, and there is reported increasing staff satisfaction and competence.
Blood component transfusion record
During the past year the Northern Ireland Transfusion Committee (NITC) has been reconvened with a new Chair, David Millar (Consultant Neonatologist, Royal Maternity Hospital).
NITC have confirmed that the National Institute for Health and Care Excellence (NICE) guideline NG24 should now be implemented throughout the region although the current Northern Ireland (NI) transfusion request form does not follow the new guidance. Due to the planned introduction of Encompass, no changes to the curent NI transfusion form are planned by the Committee.
It was agreed that in the interim period, the Trust, in order to reflect NG24 guidance, will incorporate the guidance into the Blood Transfusion Manual Policy. The Trust transfusion authorisation record has also been amended with guidance notes reflective of NG24 triggers and thresholds for blood component usage.
The Trust Transfusion Authorisation Record booklet was introduced in November 2018. The aim was to promote awareness regarding risk assessment for Transfusion Associated Circulatory Overload (TACO) and the completion of a bedside checklist prior to administration of a blood component. The booklet was updated in 2022/23 with updated illustrations regarding TACO awareness. In addition, a discharge information leaflet was attached to the booklet (the page is peforated allowing tear- off by the user). This was in keeping with recent updated guidance from Guidelines from the expert advisory committee on the Safety of Blood, Tissues and Organs (SaBTO) on patient consent for blood transfusion. “All patients who have received a transfusion have details of the transfusion (type[s] of component), together with any adverse events associated with the transfusion, included in their hospital discharge summary to ensure both the patient and their family doctor are aware. The patient should also be informed that they are no longer eligible to donate blood (with the exception of individuals who have received Convalescent Plasma from donating Convalescent Plasma to treat individuals with SARS-CoV-2).”
Haemovigilance have audited use of the Transfusion Authorisation Record following these adaptations within the Trust and specific mention was given to the Programmed Treatment Unit and Direct Assessment Unit for their excellent compliance with the completion of documentation. Deficiences were noted from all other areas including omission of patient weight documentation on the record and very poor use of the detatchable discharge patient information sheet. Haemovigilance continue to emphasise the importance of giving patients discharge information following administration of a blood component at training and education sessions. A further audit of the Trust transfusion authorisation record was planned for 2024, however due to the participation of the Haemovigilance team in a regional audit in relation to documentation regarding consent for transfusion and patient discharge information, this was paused locally. Data has been sent to the NI Transfusion Committee Audit Lead; a report will be prepared and shared with the Trust Transfusion committee in due course. As plans for digitalisation continue regionally with the implementation of Encompass, the information on the authorisation record will become accessible digitally; further audits and reports will be undertaken using data from the Encompass system.
Identification and management of sepsis within emergency departments
Sepsis is a life-threatening condition that arises when the body’s response to an infection injures its own tissues and organs. The Trust continues to monitor compliance with the Sepsis 6 bundle (oxygen, serum lactate measurement, blood cultures, intravenous (IV) fluids, antibiotics and measuring urinary output).
Within the Trust Emergency Departments, during 2023/24, overall compliance with the Sepsis 6 bundle was 13%, a decrease from 22% in 2022/23. However, when the key treatment elements of Sepsis are separated out from this the audits show that 73% of patients received antibiotics within 1 hour of identification of Sepsis, and 77% received IV fluids within 1 hour of identification of Sepsis. In order to increase awareness, compliance and performance against Sepsis 6 markers, a champion has been assigned on each site for audit and quality improvement work, which has started to increase audit activity.
During 2024/25 the Trust will continue to audit compliance with the Sepsis 6 bundle, and work with Sepsis Champions to improve compliance.
Cancer targets
The table below shows performance against the three cancer targets:
- Urgent suspected breast cancer referrals seen within 14 days
- Patients diagnosed with cancer who receive their first definitive treatment within 31 days of a decision to treat
- Patients urgently referred with a suspected cancer who begin their first definitive treatment within 62 days
| Target | 2021/22 | 2022/23 | 2023/24 | |
| 14 days | 100% | 39% | 29% | 31% |
| 31 days | 98% | 83% | 93% | 93% |
| 62 days | 95% | 40% | 35% | 36% |
The 14 day target continues to be a challenge due to an ongoing capacity gap within the Breast Service. Support to meet this demand was provided at the beginning of the year from another Trust however, this support was withdrawn mid-year due to other regional pressures. Suspect Cancer referrals for Breast increased by 14% in 2023/24 compared to the previous year.
The 31 & 62 day performance improved slightly on the previous year, however 62 day performance remains far below the target of 95%. Factors impacting 62 day performance have been continually increasing suspect cancer referrals, up 7% on previous year and 25% on 2019/20. Also delays in pathology and diagnostics, which are due to unprecedented Emergency and Red Flag demand, combined with reduced waiting list initiative funding in 2023/24.
NICE guidance
The role of the National Institute for Health and Care Excellence (NICE) is to improve outcomes for people using the National Health Service (NHS) and other public health and social care services. One way they do this is by producing evidence-based guidance and advice for health, public health and social care practitioners.
A wide range of different resources published by NICE are used by the Trust in the development and review of policies and guidelines. Examples include clinical guidelines, public health guidelines, antimicrobial guidelines, technology appraisals and clinical knowledge summaries, which are utilised by Trust staff in the development and review of Trust policies and guidelines. Once the Trust policies are approved, they are disseminated for reference by staff and are available within the Trust Policy Library.
The Department of Health (DoH) endorse the implementation, monitoring and assurance of NICE Clinical Guidelines and Public Health Guidelines. A total of 10 newly endorsed NICE Clinical Guidelines and 36 updates to previously published Clinical Guidelines were received during 2023/24. 74 NICE Technology Appraisals and 10 NICE Technology Appraisals not recommended were issued during 2023/24. No new Public health guidelines were endorsed in 2023/24.
NICE COVID-19 Rapid Guidelines have been developed in collaboration with NHS England and NHS Improvement and a cross-specialty clinical group supported by specialist societies and Royal Colleges to assist with the active management of people with suspected and confirmed COVID-19 in a number of clinical areas.
No NICE COVID-19 Rapid Guidelines were published during the 2023/24 financial year. A total of 4 updates or minor changes were received in relation to previously issued NICE COVID-19 Rapid Guidelines, as new and updated evidence, policy and practice emerged.
All such guidelines and related updates published to date have been issued to divisions within the Trust and confirmation sought regarding dissemination and implementation being taken forward, where applicable. Services identified any implementation issues.
Regular update reports were provided to relevant Committees and Groups within the Trust’s Integrated Governance and Assurance Framework Committee Structure.
International, national and regional audits
Clinical and social care audit is a way to find out if care and professional practice is in line with standards, and informs care providers and service users where a service is doing well and identifies what is not working with the aim of changing it. This allows quality improvement to take place where it is most needed and as a result improve treatment, care, safety and service quality for service users.
Trust staff have continued to engage in clinical and social care audit work, including international, national and regional audit projects.
International and national clinical or social care audit projects provide an opportunity to measure practice and services against evidence-based standards, using validated tools enabling comparison and benchmarking with other HSC Trusts and Hospitals elsewhere in the UK.
Such audits are managed or led by another organisation, with the Trust, along with other organisations, contributing to the audit. The lead organisation is responsible for reporting on the audit outcomes, however, the Trust recognises the importance of identifying learning and introducing any necessary improvements within the Trust.
During the 2023/24 financial year clinical teams contributed to a number of specific audit projects and service evaluations including:
- Acute Pharmacy and Medicines Optimisation NHS Benchmarking
- Anesthesia PraCtice for Cesarean DElivery Snapshot Study (ACCESS)
- Assessing Antibacterial and Antifungal use in ICU
- Children and Young People’s Mental Health Benchmarking
- CLAMP (Characterising leak of air in medical pneumothorax – Inspire study)
- INDEX (Interstitial Lung Disease Exacerbation – Inspire study) Managing Frailty in The Acute Setting NHS Benchmarking Network –
- Management of Bullous Pemphigoid – British Association of Dermatologists –
- Mismatch Repair in Endometrial Cancer
- PINEAPPLE Paediatric caNcellation ratEs And PerioPerative clinicaL Evaluation
- SABRE-IBD – Exploring the SAfety of perioperative Biologics for patients undeRgoing Elective surgery with Inflammatory Bowel Disease
- The Global Evaluation of Cholecystectomy Knowledge and Outcomes (GECKO) study (GlobalSurg – 4)
The Trust also contributes to National Confidential Enquiry into Patient Outcome and Death (NCEPOD) audits. NCEPOD completed during 2023/24 include:
- Testicular Torsion
- Endometriosis
- End of Life Care
These have provided an opportunity to review patient management, outcomes, safety and clinical effectiveness. A number of these audit projects are ongoing. The results generated from these audits will help inform clinical practice and improve patient care. For example, the Children and Young People’s Mental Health Benchmarking audit measures the quality of care provided against a range of evidence-based guidance. The audit engages services within mental health to measure the quality of their practice, within their model of care provision, and to trigger service improvement plans.
Regional audits
Whilst the Regulation and Quality Improvement Authority (RQIA) has stepped down its annual clinical audit programme, the Trust continues to participate in regional audits with fellow Trusts and other lead organisations. Regional audits participated in during 2023/24 include:
- Patient Discharge Information
- Consent
- The Effectiveness of the follow up pathway in children with hearing impairments with cleft palate
Additional audit programmes
In addition to participation in international, national and regional audit programmes, the Trust also has a mandatory clinical and social care audit assurance programme. The programme content is directed by audit assurances or monitoring required by external organisations and internal Trust obligations for example, learning from serious adverse incidents, complaints, litigation or to provide assurance over new policy or guidance. Clinical and social care professionals are also involved in a range of audits at service/departmental level.
The Trust’s Clinical and Social Care Audit and NICE Implementation Committee monitors’ progress with projects and the implementation of recommendations and learning following project completion. An annual clinical and social care audit report for the 2023/24 year will be produced in late 2024.








